What are the other Names for this Condition? (Also known as/Synonyms)
- Autoimmune Metaplastic Atrophic Gastritis (AMAG)
- Corpus-Restricted Atrophic Gastritis
- Type A Gastritis
What is Autoimmune Gastritis? (Definition/Background Information)
- Autoimmune Gastritis is an immune-based, non-infectious inflammation of the stomach (gastritis), as a result of antibodies (auto- or self- antibodies) produced in the body, for unknown reason
- The immune-reaction is directed against a specific type of cell, called the parietal cell or oxyntic cell, present in the mucosa/inner lining of the proximal region (upper 2/3rd) of the stomach. This immune-reaction causes destruction of the cell
- The parietal cells normally produce hydrochloric acid (famously present in the stomach contents) and a substance called ‘intrinsic factor’, which helps in absorption of vitamin B12
- Inflammatory destruction of parietal cells thus cause low hydrochloric acid (hypochlorhydria or achlorhydria), which may result in iron-deficiency anemia and deficiency of vitamin B12, leading to pernicious anemia or megaloblastic anemia
Who gets Autoimmune Gastritis? (Age and Sex Distribution)
- Autoimmune Gastritis is more common in women, than men
- A majority of individuals with the condition are older adults (commonly >60 years of age), although younger individuals (between 30-60 years) may also be affected. This disease is quite rare in those under the age of 30
What are the Risk Factors for Autoimmune Gastritis? (Predisposing Factors)
Autoimmune Gastritis risk factors are:
- A history of helicobacter gastritis (helicobacter pylori organisms may function as a trigger for subsequent production of antibodies). Chronic Atrophic Gastritis resulting from helicobacter infection may also be called, environmental metaplastic atrophic gastritis (EMAG) or ‘type B gastritis’
- Association with other autoimmune diseases, such as autoimmune thyroiditis (Hashimoto's disease), type 1 diabetes mellitus, and others like vitiligo, psoriasis, alopecia, etc.
- Family history of Autoimmune Gastritis or autoimmune diseases, in general
- Genetic predisposition with high incidence in identical twins and first degree relatives (may have association with HLA-DRB1*03 and HLA-DRB1*04)
It is important to note that having a risk factor does not mean that one will get the condition. A risk factor increases ones chances of getting a condition compared to an individual without the risk factors. Some risk factors are more important than others.
Also, not having a risk factor does not mean that an individual will not get the condition. It is always important to discuss the effect of risk factors with your healthcare provider.
What are the Causes of Autoimmune Gastritis? (Etiology)
- The cause of Autoimmune Gastritis is unknown
- The precipitating factors may include helicobacter gastritis, genetic predisposition, and other autoimmune diseases
- The immune-reaction is directed against specific cell types, called the parietal/oxyntic cell, present in the inner lining of the proximal portion of the stomach, leading to its (cell) destruction
- Specifically, the H/K-ATPase molecule on the surface of the parietal cell is the target antigen for CD4+ T cell induced immune-reaction with T-cell proliferation, production of cytokines and autoantibodies (anti-parietal cell and/or anti-intrinsic factor antibodies)
What are the Signs and Symptoms of Autoimmune Gastritis?
There is a wide range of signs and symptoms, from asymptomatic and seemingly healthy individuals, to patients presenting with severe hematological (blood-associated) and neurological disorders.
- Early clinical presentation is often insidious; consequently, the diagnosis may be missed clinically, endoscopically, and pathologically
- The symptoms may be nonspecific, such as dyspepsia (abdominal discomfort) or anemia (weakness, decreased mental concentration, headache, palpitations, pallor, etc.)
- Iron-deficiency anemia due to low hydrochloric acid is more common in Autoimmune Gastritis, than pernicious anemia due to vitamin B12 deficiency
- In some patients, severe vitamin B12 deficiency may result in neurological symptoms, such as paresthesia (tingling, burning skin sensation), gait disorders, spasticity (stiff muscles), peripheral neuropathy (dysfunction of peripheral nerves), sub-acute combined degeneration, and dementia
How is Autoimmune Gastritis Diagnosed?
The diagnosis of Autoimmune Gastritis is made by combined findings of anemia, serological (blood) studies, endoscopic findings, and gastric biopsy diagnosis.
- The anemia (low hemoglobin) may be iron-deficiency type (microcytic with low MCV) or vitamin B12 deficiency type (macrocytic with high MCV)
- Abnormal serological findings include, increased serum gastrin, reduced pepsinogen I, anti-parietal cell antibodies, anti-intrinsic factor antibodies, low serum iron, and low vitamin B12
- Endoscopically, the natural folds in the proximal stomach may appear flattened (‘atrophic’), while the distal or antral part of the stomach appears normal or unaffected. Occasionally, small nodules may be seen in the proximal stomach; which may show carcinoid tumor on biopsy
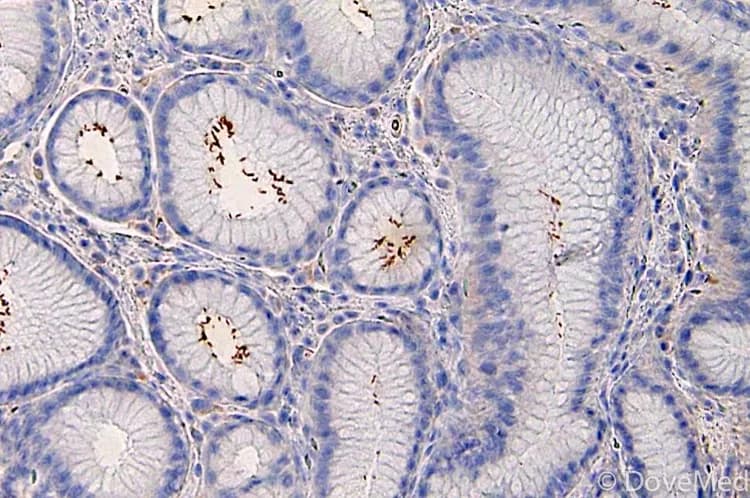
- Biopsies taken from the proximal stomach show depleted or lost parietal or oxyntic cells (atrophy), increased chronic inflammatory cells (chronic gastritis), intestinal metaplasia, and increased small clusters or hyperplasia of neuroendocrine cells (so-called ECL cells). Biopsies from distal or antral stomach show relative sparing, with no gastritis or mild gastritis
Many clinical conditions may have similar signs and symptoms. Your healthcare provider may perform additional tests to rule out other clinical conditions to arrive at a definitive diagnosis.
What are the possible Complications of Autoimmune Gastritis?
- Severe untreated Autoimmune Gastritis may lead to severe anemia and irreversible neurological symptoms
- Chronic Atrophic Gastritis carries a risk for dysplasia (pre-cancerous cellular changes) and gastric carcinoma
- Hypergastrinemia-driven ECL neuroendocrine cell hyperplasia may eventually result in small carcinoid tumors in the proximal stomach (type 1 gastric carcinoid). Such carcinoid tumors are generally benign-behaving; they are not considered aggressive, or as having significant metastatic potential
How is Autoimmune Gastritis Treated?
- Treatment of Autoimmune Gastritis concerns two separate aspects: Treatment for anemia and early detection of carcinoma and carcinoid in the stomach
- Treatment for anemia include substitution for iron and/or vitamin B12
- Early detection of carcinoma and carcinoid in the stomach involves endoscopic surveillance of gastric mucosa, optimal biopsy tissue procurement, evaluation and removal of the carcinoid nodules
How can Autoimmune Gastritis be Prevented?
- Autoimmune Gastritis cannot be prevented
- Awareness and early detection are helpful in susceptible individuals
What is the Prognosis of Autoimmune Gastritis? (Outcomes/Resolutions)
- Anemia can be successfully managed through iron and/or vitamin B12 substitution
- Early vitamin B12 substitution avoids irreversible neurological complications of severe vitamin B12 deficiency
- Gastric carcinoids develop in about 5% of patients with Autoimmune Gastritis (type I gastric carcinoid). However, these have a good prognosis with very low chances of deep invasion or metastasis
- The increased gastric carcinoma risk is still quite low, with annual incidence risk of 0.14%
- The cost-effectiveness or need for repeated endoscopic-pathological surveillance is not established, given the relatively low risk for carcinoma and aggressive carcinoids
Additional and Relevant Useful Information for Autoimmune Gastritis:
- Autoimmune Gastritis is often overlooked clinically, as well as histologically, since the symptoms and histological features may be nonspecific
- While vitamin B12 deficiency is a well-known association with Autoimmune Gastritis, iron deficiency anemia is in fact more common
- As high as 25% of patients with refractory or unexplained iron-deficiency anemia, may have clinically undetected Autoimmune Gastritis
- Normal vitamin B12 levels in the body have a physiological wide range (210-920 pg/ml); hence, in early deficiency, the vitamin B12 levels may still lie within the reference range
Related Articles
Test Your Knowledge
Asked by users
Related Centers
Related Specialties
Related Physicians
Related Procedures
Related Resources
Join DoveHubs
and connect with fellow professionals

0 Comments
Please log in to post a comment.