Researchers Visualize Brain's Serotonin Pump, Provide Blueprint For New, More Effective SSRIs
Researchers at Oregon Health & Science University's Vollum Institute have uncovered remarkably detailed 3-D views of one of the most important transporters in the brain -- the serotonin transporter. Their study, published online today in the journal Nature, provides fresh insight into how citalopram and paroxetine, two of the most widely prescribed selective serotonin reuptake inhibitors, or SSRIs, interact with and inhibit serotonin transport.
Visualizing this molecular structure creates a platform for designing new, more effective small molecule therapeutics to treat depression and anxiety.
"The heavy toll that devastating illnesses like anxiety and depression have on families and communities is, in many ways, incalculable. Revealing the precise structure of the serotonin transporter holds tremendous promise for the development of life-changing drug treatments for these diseases," said Eric Gouaux, Ph.D., senior scientist in the Vollum Institute at OHSU, National Academy of Sciences member, and Howard Hughes Medical Institute Investigator. Gouaux is an internationally recognized crystallographers in the area of neurotransmitter receptor and transporter structure.
Influencing virtually all human behaviors, serotonin regulates the activity of the central nervous system as well as processes throughout the body, from cardiovascular function to digestion, body temperature, endocrinology and reproduction. The serotonin transporter acts as a molecular pump for serotonin, recycling the neurotransmitter following neuronal signaling. Serotonin shapes neurological processes including sleep, mood, cognition, pain, hunger and aggression.
When SSRIs were first developed in the 1980s, the molecular identity of the transporter they targeted was unknown. In the 1990s, researchers realized that SSRIs had a common target -- the serotonin transporter. By binding to the transporter and blocking serotonin from being taken back up by the transporter into the cell, SSRIs allow serotonin to remain outside of the cell longer than normal, potentially prolonging neuronal signaling. Though SSRIs are widely used to treat anxiety and depression, the molecular mechanism by which they block the transporter is not fully understood.
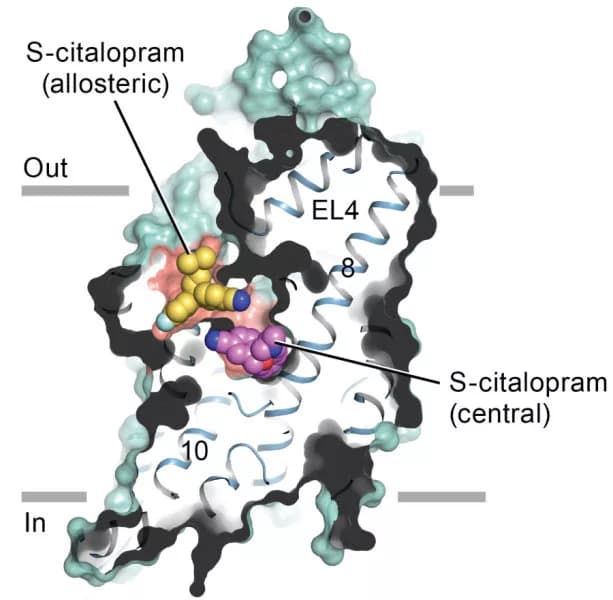
Gouaux's lab used X-ray crystallography to capture images of the transporter. In this paper, they report that SSRIs lock the transporter in an outward-open conformation by loading in the central binding site, directly blocking serotonin binding.
The study, "X-ray structures and mechanism of the human serotonin transporter," was authored by Gouaux; Jonathan A. Coleman, Ph.D.; and Evan M. Green, a research assistant who is now a Ph.D. student at University of California San Francisco.
This work was supported by Bernie and Jennifer LaCroute and the National Institutes of Health (NIH) (5R37MH070039). Coleman has support from a Banting postdoctoral fellowship from the Canadian Institutes of Health Research.
The above post is reprinted from materials provided by Oregon Health & Science University. Note: Materials may be edited for content and length.
Disclaimer: DoveMed is not responsible for the adapted accuracy of news releases posted to DoveMed by contributing universities and institutions.
Primary Resource:
Coleman, J. A., Green, E. M., & Gouaux, E. (2016). X-ray structures and mechanism of the human serotonin transporter. Nature.
Related Articles
Test Your Knowledge
Asked by users
Related Centers
Related Specialties
Related Physicians
Related Procedures
Related Resources
Join DoveHubs
and connect with fellow professionals

0 Comments
Please log in to post a comment.