Encouraging Results For Patients With Aggressive Brain Cancer
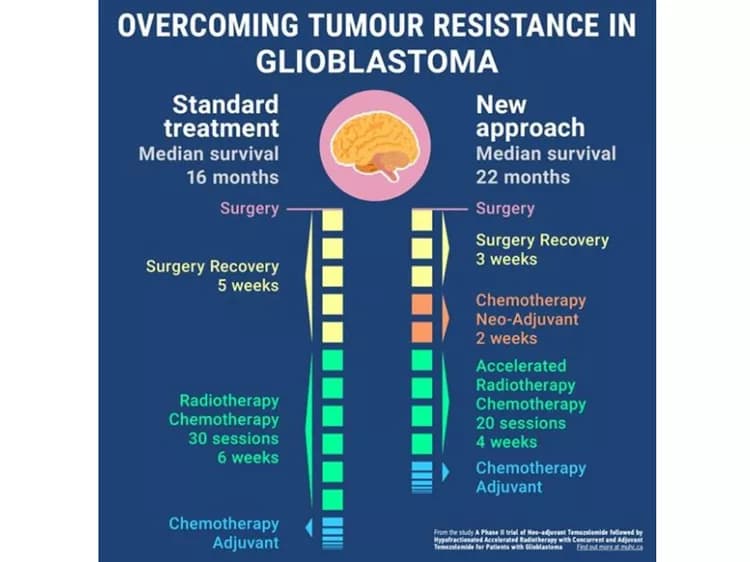
Being diagnosed with a malignant brain tumor is devastating news for patients and their loved ones. Whereas some types of tumor respond well to treatment, others such as glioblastomas -- the most common and aggressive brain tumors -- are known to recur and progress within short times from the diagnosis. Patients diagnosed with this type of cancer, and who undergo current standard treatment, have a median survival of 16 months.
Based on recent information on the mechanisms of chemotherapy, a team of researchers of the McGill University Health Centre (MUHC) developed a new clinical approach to increase the efficiency of treatment in glioblastomas that increased the median survival to 22 months -- bringing much needed hope to those affected by this aggressive disease. The findings of this promising phase II clinical trial have been published in the International Journal of Radiology Oncology.
"Glioblastomas are very difficult to treat," explains Dr. George Shenouda, radio-oncologist at the MUHC and lead author of the study. "These tumors grow and spread quickly throughout the brain, making it very difficult to completely remove with surgery."
The standard treatment for glioblastomas consists of removing as much of the tumor as possible with surgery and then eliminating what is left through radiotherapy combined with chemotherapy. After surgery, patients need at least 4-5 weeks of recovery before starting radiotherapy. Unfortunately, during this recovery time any remaining cancer cells will continue to grow. To make matters more complicated, the remaining cancer cells, mainly cancer stem cells, can be more resistant to radiotherapy and chemotherapy.
With their new approach, the team of researchers added chemotherapy prior to radiotherapy, also called neo-adjuvant chemotherapy. Giving neo-adjuvant chemotherapy prevented the tumor from progressing during recovery and increased the patients' survival.
After the neo-adjuvant chemotherapy, the patients were then given accelerated radiotherapy. "We had better control over the tumor by giving patients the same overall dose of radiotherapy in fewer sessions and a shorter period of time. By doing this, we increased the efficacy of the treatment and we believe that in turn the treatment targeted the stem cells, which are the basis of recurrence. Reducing the radiotherapy sessions by one-third also alleviates the burden for patients. In addition, this represents a considerable cost reduction of delivery of treatment" says Dr. Shenouda.
Although additional research is required, the initial results are very promising. "Fifty per cent of the patients in our study have survived two years since their diagnosis -- this is very encouraging and we are very positive about the outcome," says Dr. Shenouda.
Materials provided by McGill University Health Centre. Note: Content may be edited for style and length.
Disclaimer: DoveMed is not responsible for the accuracy of the adapted version of news releases posted to DoveMed by contributing universities and institutions.
Primary Resource:
Shenouda, G., Souhami, L., Petrecca, K., Owen, S., Panet-Raymond, V., Guiot, M. C., ... & Abdulkarim, B. (2017). A Phase 2 Trial of Neoadjuvant Temozolomide Followed by Hypofractionated Accelerated Radiation Therapy With Concurrent and Adjuvant Temozolomide for Patients With Glioblastoma. International Journal of Radiation Oncology* Biology* Physics, 97(3), 487-494. DOI: 10.1016/j.ijrobp.2016.11.006
Related Articles
Test Your Knowledge
Asked by users
Related Centers
Related Specialties
Related Physicians
Related Procedures
Related Resources
Join DoveHubs
and connect with fellow professionals

0 Comments
Please log in to post a comment.